Bicillin L-a
Benzylpenicilline Benzathine
CAS: 41372-02-5
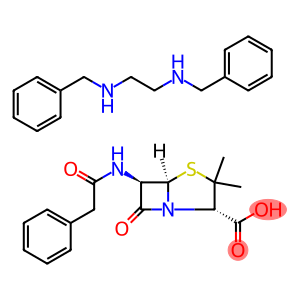
Molecular Formula: C48H64N6O12S2
Bicillin L-a - Names and Identifiers
| Name | Benzylpenicilline Benzathine |
| Synonyms | Permapen Bicillin Bicillin l-a Bicillin L-a Penicillin G benzathine BENZYLPENICILLIN BENZATHIN Benzylpenicilline Benzathine Penicillin G benzathine [usan] Penicillin G Benzathine [USAN] Penicillin G Benzathine (200 mg) Penicillin G benzathine tetrahydrate BENZATHINE PENICILLINE G TETRAHYDRATE PENICILLIN G BENZATHINE, TETRAHYDRATE) N,N'-dibenzylethane-1,2-diamine,(2S,5R,6R)-3,3-dimethyl-7-oxo-6-[(2-phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylicacid,tetrahydrate |
| CAS | 41372-02-5 |
| EINECS | 804-898-2 |
| InChI | InChI=1/2C16H18N2O4S.C16H20N2.4H2O/c2*1-16(2)12(15(21)22)18-13(20)11(14(18)23-16)17-10(19)8-9-6-4-3-5-7-9;1-3-7-15(8-4-1)13-17-11-12-18-14-16-9-5-2-6-10-16;;;;/h2*3-7,11-12,14H,8H2,1-2H3,(H,17,19)(H,21,22);1-10,17-18H,11-14H2;4*1H2/t2*11-,12+,14-;;;;;/m11...../s1 |
Bicillin L-a - Physico-chemical Properties
| Molecular Formula | C48H64N6O12S2 |
| Molar Mass | 981.18 |
| Melting Point | >118°C (dec.) |
| Boling Point | 1299.8°C at 760 mmHg |
| Flash Point | 739.9°C |
| Solubility | DMSO (Slightly), Methanol (Slightly) |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | neat |
| Color | White to Off-White |
| Storage Condition | Refrigerator |
| Use | Suitable for penicillin sensitive bacteria caused by mild or moderate infections, such as Pneumonia, scarlet fever, tonsillitis, otitis media, gonorrhea, etc |
| In vivo study | Penicillin G benzathine tetrahydrate displays activity and antibacterial effect in CD-I mice infected with Staphylococcus aureus Smith. |
Bicillin L-a - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 42/43 - May cause sensitization by inhalation and skin contact. |
| Safety Description | S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S24/25 - Avoid contact with skin and eyes. |
| HS Code | 29411099 |
Bicillin L-a - Reference
| Reference Show more | 1. Wang Xiaomin LV Ruina Huang Jing Li Changtian. Effects of different drying methods on quality and polyphenol oxidase activity of Flammulina velutipes [J]. Science and Technology of food industry, 2019, 40(22):77-81. 2. Sun Jing-Chen. Study on ultrasonic treatment to improve the solubility and emulsifying properties of rice bran protein [J]. Agricultural products processing, 2019, No.484(14):39-44 48. 3. Li, Li, Feng, Li, Wang, Guangqiang, et al. Extraction, preparation and purification of Dava gum [J]. Journal of Food and Fermentation Technology, 2019, 055(004):52-58. 4. Jiang Hongbo, Ding baomiao, Zhang Xin. Adsorption Kinetics of macroporous resin on rinsed water of black carp surimi [J]. Food industry, 2019(6):203-206. 5. Zhang Xin, Ding baomiao, Jiang Hongbo. Study on the adsorption thermodynamics of macroporous resin on the washing water of black carp surimi [J]. Food Research and Development, 2019, 40(04):51-54. |
Bicillin L-a - Standard
Authoritative Data Verified Data
(2S,5R,6R)-3, 3-dimethyl-7-oxo-6-(2-phenylacetamido)-4-thia-1-azabicyclo [3. 2. 0] N ,AT-dibenzylethylenediamine salt tetrahydrate of heptane-2-carboxylic acid, or a sterile powder prepared by adding appropriate buffer and suspending agent. According to the calculation of anhydrous, dibenzylethylenediamine (C16H20N2) should be 24.0% ~ 27.0%, containing penicillin (C16H18N204S) should be 69.9% ~ 75.0%, per 1 mg containing penicillin should be 1244~1335 units.
Bicillin L-a - Trait
Authoritative Data Verified Data
- This product is a white crystalline powder.
- This product is soluble in N ,N-dimethylformamide or formamide, slightly soluble in ethanol, and slightly soluble in water.
Bicillin L-a - Differential diagnosis
Authoritative Data Verified Data
In the chromatogram recorded under the content determination item, the retention time of the two main peaks of the test solution should be consistent with the retention time of the corresponding two main peaks in the control solution.
Bicillin L-a - Exam
Authoritative Data Verified Data
pH
take 50mg of this product, Add 10ml of water to make suspension, according to the law (General 0631),pH value should be 5.0~7.0.
Related substances
new system for clinical use. Accurately weigh about 70mg of this product, put it in a 50ml measuring flask, add 10ml of acetonitrile, shake to disperse evenly, and then add 10ml of methanol to fully shake to dissolve, immediately dilute to the scale with phosphate buffer (take potassium dihydrogen phosphate 6.8g and dipotassium hydrogen phosphate 1.14g, add water to dissolve and dilute to 1000ml), shake, as a test solution; Take lml with precision, in a 100ml measuring flask, dilute to the scale with the above phosphate buffer, and shake as a control solution. According to high performance liquid chromatography (General 0512) determination, with eighteen alkyl silane bonded silica gel as filler (end group tail sealing); mobile phase A was 0.05mol/L potassium dihydrogen phosphate solution (adjusted to pH 3.1 with phosphoric acid), and mobile phase B was Methanol. The column temperature was 40°C; the detection wavelength was 220mn. Take 70mg of this product, put it in 50ml measuring flask, add 10ml of acetonitrile and 5ml of methanol to dissolve, add 0.1 mol/L hydrochloric acid solution 2.0ml, 10 minutes, add 0.1 mol/L sodium hydroxide solution 2.0ml neutralization, dilute to the scale with 0.05mol/L sodium hyaluronate buffer (pH 6.0), shake well, as the system applicable solution, take 20u1 injection human liquid chromatograph, record chromatogram, the retention time of the penicillin peak is about 20 minutes, and the degree of separation of the dibenzylethylenediamine peak from the adjacent impurity bees and the degree of separation of the penicillin peak from the adjacent impurity peaks should meet the requirements. 20 u1 of the test solution and the control solution were respectively injected into the human liquid chromatograph, and the chromatograms were recorded. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be more than 2 times (2.0%) of the area of the two main peaks of the control solution, the sum of each impurity peak area shall not be greater than 3.5 times (3.5%) of the sum of the two main peak areas of the control solution, and the peaks smaller than the two main peak areas and 0.05 times in the chromatogram of the test solution shall be ignored.
moisture
take this product, according to the moisture determination method (General 0832 first method 1), the moisture content should be 5.0% ~ 8.0%.
aspiration test
take this product l.Og, add water 4ml, shake, with a syringe with a 5.5 needle, should be able to pass smoothly, not blocked. Visible foreign matter 5 parts of the maximum specification under the preparation item, respectively, plus N ,N-dimethylformamide appropriate amount of dissolution, according to the law inspection (General 0904), should comply with the provisions. (For aseptic dispensing)
bacterial endotoxin
take this product, check according to law (General 1143), per 1000 units of penicillin containing endotoxin should be less than 0.25 EU. (For injection)
sterile
take this product, use appropriate solvent to disperse evenly, add penicillinase inactivation, check according to law (General rule 1101), should comply with the provisions. (For aseptic dispensing)
Bicillin L-a - Content determination
Authoritative Data Verified Data
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
silica gel bonded with octa-alkyl silane (end-capped) was used as the filler; 0.05mol/L potassium dihydrogen phosphate solution (adjusted to pH 5.1 with phosphoric acid)-acetonitrile (83:17) was used as the mobile phase; the detection wavelength was 220nm. The system applicable solution 20u1 under the item of related substances is injected into the human liquid chromatograph, and the chromatogram is recorded. The separation degree between the dibenzylethylenediamine peak and the adjacent impurity peak and the separation degree between the penicillin peak and the adjacent impurity peak shall meet the requirements.
assay
accurately weigh about 35mg of this product, put it in a 50ml measuring flask, Add 10ml of acetonitrile to shake to make uniform dispersion, and then add 10ml of methanol to fully shake to dissolve, immediately dilute to the scale with phosphate buffer under the item of related substances, shake well, as a test solution, take 20u1 injection liquid chromatograph, record chromatogram; Take benzathine penicillin reference substance, same method determination. The content of penicillin (C16H18N204S) and dibenzylethylenediamine (Cl6H20N2) in the sample was calculated by peak area according to external standard method. Each 1 mg of C16H18N204S is equivalent to 1780 penicillin units.
Bicillin L-a - Category
Authoritative Data Verified Data
lactam antibiotics, penicillins.
Bicillin L-a - Storage
Authoritative Data Verified Data
sealed and stored in a dry place.
Bicillin L-a - Benzathine penicillin for injection
Authoritative Data Verified Data
This product is a sterile powder made from the dibenzylethylenediamine salt of penicillin with appropriate buffer and suspending agent. According to the calculation of anhydrous matter, containing dibenzylethylenediamine (C16H2ON2) should be 24.0% ~ 27.0%, containing penicillin (C16H18N204S) should be 69.9%-75.0%, per 1 mg containing penicillin should be 1244~1335 units; the content of penicillin (C16H18N204S) shall be between 95.0% and 105.0% of the labeled amount based on the average loading.
trait
This product is a white crystalline powder.
identification
In the chromatogram recorded under the content determination item, the retention time of the two main peaks of the test solution should be consistent with the retention time of the corresponding two main peaks in the control solution.
examination
- needle test take 1 bottle of this product, 1 ml of water per 300,000 units, shake well, and use a syringe with a 5.5-gauge needle to draw, which should pass smoothly without blocking.
- visible foreign matter 5 bottles of this product, respectively, plus N,N-dimethylformamide appropriate amount of dissolution, according to law inspection (General rule 0904), should comply with the provisions.
- the pH value, related substances, moisture, bacterial endotoxin and sterility shall be checked according to the method under the item of benzathine penicillin, and shall be in accordance with the regulations.
- others should comply with the relevant provisions under injection (General 0102).
Content determination
The content under the item of loading amount difference was obtained by measuring according to the method of benzathine penicillin. Each LG of C16H18N20 4S corresponds to 1780 penicillin units.
category
Same as benzathine penicillin.
specification
(1) 300,000 units (2) 600,000 units (3) 1.2 million units
storage
sealed and stored in a dry place.
Supplier List
CAS: 41372-02-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 41372-02-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 41372-02-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 41372-02-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 41372-02-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 41372-02-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025



